Urologic Surgery
Latest News
Latest Videos

CME Content
More News

"Fake news rules the world today. It certainly applies to the use of mesh in female pelvic health," writes Gopal H. Badlani, MD.

Although mesh has been banned for prolapse repair, midurethral slings remain a crucial treatment option for stress urinary incontinence.

"All we can do as physicians is reach out to patients with dignity and respect for who they recognize themselves to be," says one urologist.

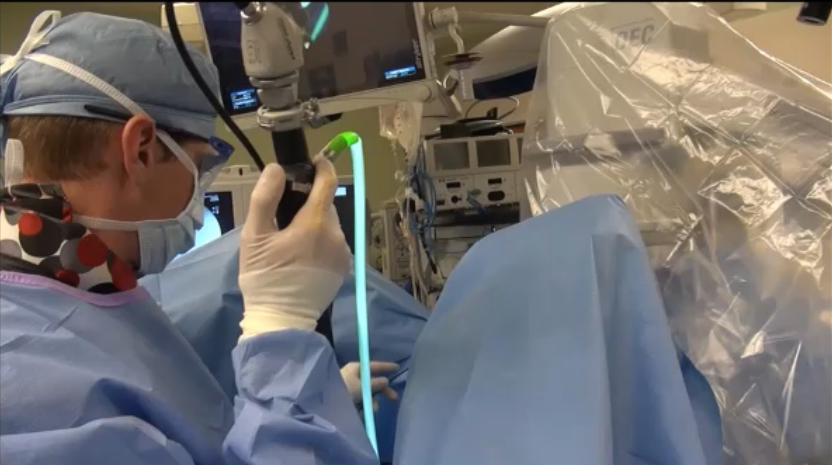
This article highlights some of the emerging endoscopic robotic systems in urology.

In this article, we provide an overview of the key concepts a clinician must take into consideration when consulting with men interested in undergoing a cosmetic surgical procedure.

Evidence suggests fallopian tube removal will lead to reduced ovarian cancer incidence.

In this Urology Times Q&A, Jessica Nelson, MPAS, PA-C, past president of the Urological Association of Physician Assistants, offers a physician assistant’s perspective on PAs performing urologic procedures, PA training, and more.

Also read about expansions to an intermittent catheter line, a new radiofrequency treatment for improving local vaginal circulation, an app for men navigating fertility challenges, and more.

Research on a cranberry supplement for decreasing UTI incidence as well as a finding that ketorolac was more effective than narcotics in controlling renal colic were among the AUA 2018 take-homes in quality improvement and patient safety.

“A decade ago it was rare to have advanced practice providers as part of routine urologic care. Now they’re integral and a necessary part of any active urologic practice,” says Bradley A. Erickson, MD, MS.

Reconstructive surgery for complications from female-to-male genitourinary reconstruction surgery and complex reoperative hypospadias repair were among noteworthy trauma/reconstruction topics at AUA 2018.

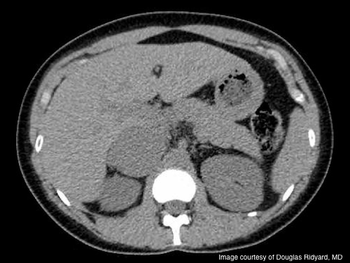
An 18-year-old previously healthy collegiate athlete presented to the emergency room with a rapid weight gain of 20 pounds during the previous 2 months. She complained of significant fatigue and irregular menstrual periods.

Research examining transcutaneous electrical nerve stimulation for nocturnal enuresis as well as pediatric sacral nerve stimulator explantation rate due to complications were among the noteworthy pediatric urology studies from AUA 2018.

Other take-home messages on transplantation from AUA 2018 included the finding that kidney paired donations have more than doubled since their inception in 2009 as well as early robotic kidney transplantation studies.

These videos demonstrate significant advances in percutaneous nephrolithotomy technique that can serve to optimize outcomes, reduce OR time, and help achieve optimal results.

Noteworthy minimally invasive surgery research from AUA 2018 also included topics such as robot-assisted versus open radical cystectomy as well as minimally invasive retroperitoneal lymph node dissection in men with testis cancer.

A mobile app for stone metaphylaxis and an algorithm defining specific indications for stent omissions were among the other highlights in endourology/stones at AUA 2018.

Recent studies point to the significant potential activity trackers hold in urology.

Few urology residents receive didactic education on gender-affirming care, survey shows.

"The world of laser vaginal rejuvenation is fast paced, with new companies adding a laser platform nearly each month," write Jessica DeLong, MD, and Jennifer Miles-Thomas, MD.

Recent data support the product’s use in protecting organs at risk for radiation exposure.

Laurence Baskin, MD, presents the case of a baby boy who had a tough forceps delivery, with accompanying sonographic images.

"Personally, I’ve tried to use a lot more NSAIDs and over-the-counter pain medications but there are times where I give very short-course Percocet or Norco to cover a patient for surgery," says one urologist.

This article examines current developments in robotic urologic surgery and future directions, including the introduction of alternative surgical robots.