
Test your knowledge of this quiz on the Canadian Urological Association's recommendations for germline genetic testing in prostate cancer management.

Test your knowledge of this quiz on the Canadian Urological Association's recommendations for germline genetic testing in prostate cancer management.
"Overall, the take-home message is that in patients with BCG-unresponsive non–muscle invasive bladder cancer, you can consider nadofaragene as an option among those that are available for treating these patients," said Vikram Narayan, MD.

The clearance is supported by data from the PRESERVE trial, which evaluated the safety and effectiveness of the NanoKnife System in patients with intermediate-risk prostate cancer.

"We found that the majority of malpractice claims were filed in response to delayed diagnosis or treatment and no cases were filed in response to surveillance practices or surgical techniques," said Adri M. Durant, MD.

CR rate was 98% (39/40) at 3 months, 94% at 6 months (34/36), and 81% at 12 months (21/26).

The priority review decision is based on results of the phase 3 NIAGARA study (NCT03732677).

As centrally assessed, the CR rate was 83.5% (95% CI, 73.9-90.7); the CR rate was 85.9% (95% CI, 76.6-92.5) per investigator assessment.
At month 6, the overall high-grade complete response rate was 72% (13 of 18
The overall complete response rate was 74.5% (95% CI, 65.4%-82.4%).
Anemia was the most frequently observed any-cause AE and was seen in 485 (84.2%) patients.

PT217 is currently under investigation in the phase 1/2 SKYBRIDGE trial in patients with neuroendocrine cancers.

Overall median time from first documented biomarker test date to index date was 5.5 months.

"That pan-tumor approval and this patient case really highlight that in prostate cancer, we should be checking HER2 by immunohistochemistry," says Maneesh R. Jain, MD.
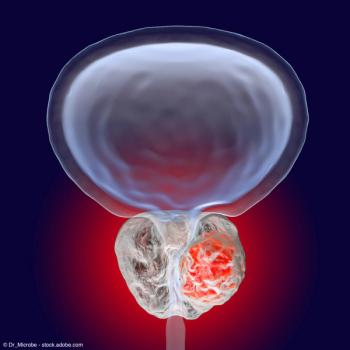
Data showed that all patients achieved a PSA decline of at least 50%.
Ferring has announced the opening of the first clinical trial sites for 2 studies within the ABLE clinical trial program as well as the launch of the phase 1/2 LUNAR trial.

“The combination of pembrolizumab and chemotherapy presents a promising new treatment approach for these challenging-to-treat, rare cancers and could be a major breakthrough for patient care,” says Arnold I. Chin, MD, PhD.

"Most of the published data about HIFU tended to be with relatively short-term follow-up," says Ralph J. Miller Jr, MD.
The phase 2 CELLVX-230 trial is exploring the potential of FK-PC101 to delay or prevent prostate cancer recurrence following prostatectomy.

"The final rule for 2025 poses several challenges to urology. Lobbying efforts to preserve telehealth access and the CF will continue, perhaps dragging into 2025," write Jonathan Rubenstein, MD, and Mark Painter.
“Our study highlights the potential of epigenomic profiling from a simple blood draw to provide a real-time, non-invasive readout of PSMA expression that corresponds with treatment response," says Jacob E. Berchuck, MD.

“The median DOR of 47.8 months in patients who achieved complete response with JELMYTO provides evidence of robust durability in maintaining control of low-grade UTUC over an extended period,” says Phillip Pierorazio, MD.

The study showed significant reductions in both anxiety and depressive symptoms following the intervention.

Data from the CAPItello-281 trial also showed a trend toward an improvement in overall survival.

The trial is evaluating the diagnostic performance of copper Cu 64 PSMA I&T PET/CT in men with suspected biochemical recurrence of prostate cancer following treatment with curative intent.

In the recent news release, Koelis reported that the final patient was enrolled in the VIOLETTE study in September 2024.

The sNDA is supported by data from the pivotal phase 3 ARANOTE trial.

“Prostate cancer patients with life expectancies of less than 5 or 10 years were being subjected to treatments that can take up to a decade to significantly improve their chances of surviving cancer, despite guidelines recommending against treatment," says Timothy J. Daskivich, MD.
Initial results on ADI-270 are anticipated for the first half of 2025.

At the time of data cutoff, 71% of patients achieved a complete response following treatment.

The primary objective is to compare the detection rate of sites of prostate cancer recurrence between the 2 agents.