
Anti-tumor activity was observed across all patients with FGFR3-altered metastatic urothelial carcinoma who were treated at the dose levels of 90 mg and higher.

Anti-tumor activity was observed across all patients with FGFR3-altered metastatic urothelial carcinoma who were treated at the dose levels of 90 mg and higher.
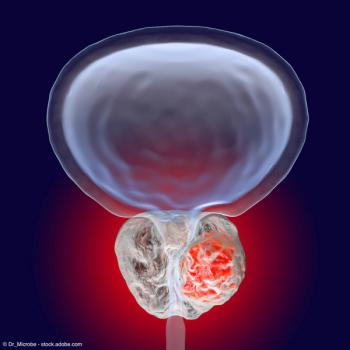
The NDA submission is supported by data from a single-arm, phase 3 trial of leuprolide mesylate administered as 2 injections, 3 months apart.

The RMAT designation is supported by interim data from the ongoing phase 1 TRAVERSE trial.

“The majority (76%) of patients not yet meeting the Phoenix criteria did have PSMA-avid lesions on PSMA-PET, suggesting [prostate cancer] recurrence," says Evelien J E van Altena.
The phase 3 ENVISION trial met its primary end point by demonstrating a 79.6% complete response rate at 3 months following the first instillation of the therapy.

“These findings support the safety of vaginal estrogen for treatment of local menopause symptoms, even in high-risk women,” says Gabriella M. Rustia, MD.

The approval is supported by data from the phase 3 SURE 1 and REASSURE trials.

The therapeutic response rate was 79% at 24 months, which is comparable to the rates seen at 6 months (78%) and 12 months (82%).

At 1 month, the mean decrease in the OAB-Q symptom severity subscale score was -31.2 in the 1-injection cohort vs -39.5 in the 10-injection cohort.

Patients with higher baseline HEI and E-HEI scores were less likely to experience grade reclassification.

The cystoscopy solution was granted a CE mark for regulatory clearance in Europe in October 2023.

The trial is comparing the diagnostic performance of 61Cu-NODAGA-PSMA I&T with that of 18F-piflufolastat.

The codes will go into effect on January 1, 2026.

None of the videos mentioned the long-term impact of exogenous testosterone on spermatogenesis.

The decision to withdraw sacituzumab govitecan from the market was made in consultation with the US FDA following negative data from the phase 3 TROPiCS-04 trial.

Almost all studies (93.5%) reported an improvement in validated patient-reported outcomes following treatment.

SMSNA 2024 abstract data show shockwave therapies and platelet-rich plasma, either alone or in combination, may be promising treatments for ED.

68.8% of patients had at least a 30% improvement in ventral curvature from baseline.

“The take-home message…is it doesn't matter whether you put the reservoir in the ectopic location or the prevesical location. Patients tend to be satisfied in both locations,” said Mohit Khera, MD, MBA, MPH.

The investigators reported a significantly greater prevalence of ED in men with BPH (P < .0001).

“Allowing these women to have a fulfilling life after cancer is one of the best things we can do as physicians,” says Talia A. Helman, MD.

For the survey question “Do 5-ARIs reduce serum PSA?” 8 (9.4%) respondents said “no” and 25 (29.4%) said “not sure.”

Data from a Mayo Clinic study show, despite some reports of discomfort and dissatisfaction, a majority of patients receiving penile prosthesis are satisfied 1 year in.

New research better defines the link between cardiovascular disease risk and prevalence of ED per serum ceramide levels.

Data from a new SMSNA 2024 abstract show an association between significantly high BMI and risk of azoospermia and oligospermia.

Following use of MED3000, the investigators reported significant improvements in all 5 SEP questions in both patients and partners over the course of the 12 weeks.

The investigators reported a significant improvement in IPSS, from 21.9 at baseline to 12.6 at 4 years.

“Initiating oral TU therapy with Kyzatrex at 400 mg BID is safe and effective in achieving therapeutic serum testosterone levels," wrote the authors.

Overall, 4 of 5 patients with low-grade disease who received bel-sar with light activation achieved a complete clinical response.

The investigators found that only 2 (1.4%) urology programs and 12 (5.3%) OB/GYN programs included female sexual dysfunction content.