The BLA for TLX250-CDx was initially submitted to the FDA in June 2024.

Catch up on all the notable drug and device approvals in urology over the past year.

"If a practice has determined in writing that there are insurers whose policies advised that they will not accept this code, it seems reasonable not to report that code to that insurer even when criteria are met," write Jonathan Rubenstein, MD, and Mark Painter.
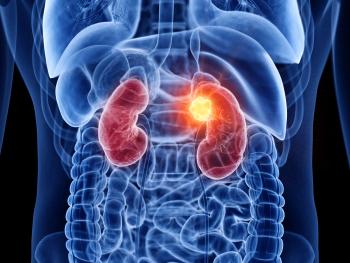
As the year comes to a close, we revisit some of this year’s top content on the management of renal cancer.
As the year comes to a close, we revisit some of this year’s top content on prostate cancer.

The approval is supported by data from the phase 3 CheckMate-67T trial.

"As a result of the changes and the programmed phase-in of the changes, the cystoscopy code total RVU for the office setting will be 8% lower in 2025 than the current value," write Jonathan Rubenstein, MD, and Mark Painter.
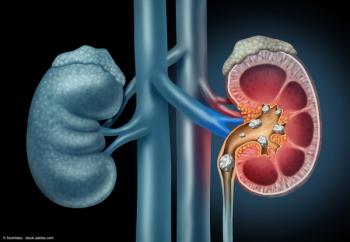
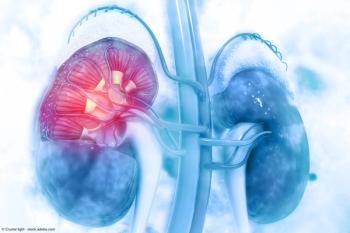
As the year comes to a close, we revisit some of this year’s top content on kidney stone management.

Test your knowledge of these often-overlooked but essential aspects of genetic and genomic testing.

The SURE procedure also demonstrated significantly better outcomes on the study’s secondary end points of stone clearance and residual stone volume.

"I hope that [attendees] will be exposed to a community where they feel that they're able to freely ask questions, gain accurate information, and feel heard, validated, and connected," says Lauren E. Corona, MD.

The alert was issued to clarify that the devices should not be used for suction and irrigation.

The investigators found a positive correlation between PSA levels and SUVmax.
“Our findings highlight the coordinated interplay between GCN2 and p53 regulation during nutrient stress and provide insight into how they could be targeted in developing new therapeutic strategies for [prostate cancer]," write the authors.

Initial data from the trial are expected to be released in the first half of 2025.

The redePHine trial is expected to launch in the first half of 2025.

The inclusion of the test is supported by more than 25 studies demonstrating its clinical utility, including 2 studies published earlier this year.

As the year comes to a close, we revisit some of this year’s top content on next-generation imaging agents in urology.

Test your knowledge of this quiz on the Canadian Urological Association's recommendations for genomic profiling of tumors in prostate cancer.

Pluvicto was approved by Health Canada in August 2022.

"I think this is a very important trial, just to bring some importance about patients' worries about being sexually active with urinary incontinence," says LeRoy Jones, MD.

From baseline to 1 year, the Ki-67 index decreased in the dietary intervention cohort by 15% and increased in the control cohort by 24%.

Age, race, and hormonal contraceptive use did not significantly impact the efficacy of sildenafil cream, 3.6%, in female sexual arousal disorder.
A final decision on marketing authorization is anticipated for Q1 of 2025.
“If approved, this approach has the potential to transform the treatment paradigm in prostate cancer, offering patients with localized disease an effective treatment option that may reduce the risk of disease recurrence," says Glen Gejerman, MD, MBA.

The primary aims for the EVANESCE-II trial are to confirm the safety of the FemPulse System and to establish noninferiority to first-line tolterodine.

Specifically, 90% of patients in the HIFU arm vs 86% of patients in the radical prostatectomy arm were free from subsequent definitive treatment at 30 months.

Cretostimogene grenadenorepvec is an investigational oncolytic immunotherapy for patients with BCG-unresponsive high-risk NMIBC.
"The durability response for these patients was impressive given the fact that these patients were those who, by definition and by inclusion criteria, were recurrent within a year at [baseline]," says Sandip M. Prasad, MD.